Human IgG / IgM Library Primer Set
- Human IgG and IgM Library Primer Set 1 and 2
- 16 vials per primer set
Product description
| Quantity | 2x16 primers |
|---|---|
| Purification | HPLC or HPSF purification |
| Storage | -20°C |
| Intended use | Research use only |
| Formulation | Ready-to-use for common PCR (concentration 10 pmol/µl in 10 mM Tris-HCl pH 8, 1 mM EDTA) |
| Purity | HPLC or HPSF purification |
| Specificity | PCR amplification (Set 1) and cloning (Set 2) of human IgG and IgM heavy and light chain variable domain coding regions |
| Stability | Minimum 1 year at -20°C |
| Product description | Generation of large repertoires of rearranged immunoglobulin variable domain coding regions for the construction of human IgG and IgM scFv-antibody libraries. Amplification of immunoglobulin variable gene fragments from single B cell clones. |
| Note | Volume: 20 to 230 µl each |
Background
Introduction
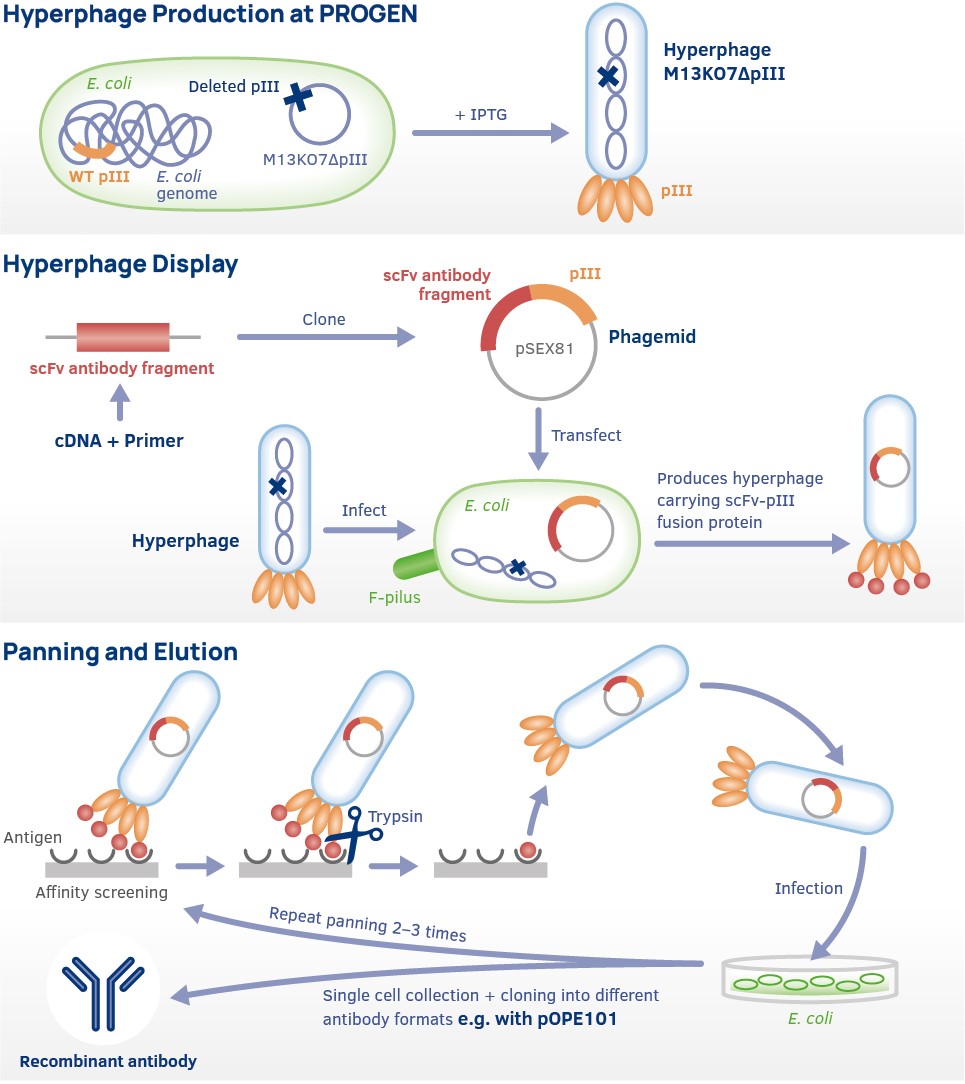
Many monoclonal antibodies of animal origin are valuable diagnostic agents. Their therapeutic use, however, has been frequently limited by hypersensitivity reactions or by antiimmunoglobulin responses. The replacement of the animal with human antibodies solves most of these problems, but the production of human monoclonal antibodies by classical hybridoma techniques or EBV transformation is limited by the instability of cell lines, low antibody yields and the ethic limitations of immunizing humans with antigens. A promising alternative to the hybridoma technology is the production of recombinant antibodies. Pioneering work of the last decade showed that it is possible to amplify rearranged immunoglobulin genes from B-lymphocytes, to insert them into different vectors, and to express them in bacteria, in yeast, in insect, mammalian or plant cells. Moreover, the randomized combination of cloned heavy and light chain immunoglobulin gene fragments allowed the construction of human antibody libraries. These libraries enable the isolation of specific antibodies against particular antigens by phage display techniques.
This screening strategy offers the possibility to obtain specific human antibodies without prior immunization of a lymphocyte donor. One prerequisite for generating highly diversified human antibody libraries, however, is the development of PCR primers capable of amplifying all rearranged immunoglobulin genes. Previous primer designs have been based on nucleotide sequences of immunoglobulin genes. PROGEN offers a primer set based on amino acid sequences of antibodies, which allows the amplification of rearranged human immunoglobulin genes of individual B cell clones as well as of larger B cell populations for the construction of human scFv antibody libraries.
PCR Primer Design
The PCR oligonucleotide primers were designed for the amplification of human rearranged IgG and IgM variable domain coding regions in a two step PCR. The first set of PCR primers (Set 1) leads to the amplification of the antibody genes from a lymphocyte cDNA source. The forward primers correspond to the first 1-9 amino acid residues of the variable immunoglobulin domains (FR1), the reverse primers to the amino acids 115-121 of CH1, to 109-116 of CL-kappa or to 115-122 of CL-lambda. The primer design started from amino acid consensus sequences of immunoglobulins as described by Kabat et al. (1991), which were then deduced from the codon usage preferentially used in human immunoglobulin variable domain coding regions (Kabat et al., 1991). To allow sequence variability, wobble nucleotides were introduced in certain positions to account for the percentage of occurence of the encoded amino acids as represented in the Kabat database. The primers correspond to the subgroup specification based on the amino acid sequence similarities, as classified by Kabat et al. (1991). However, since both the kappa1, kappa4 and the lambda2, lambda5 variable light chains are highly homologous, only one primer was designed for each group (1F for kappa1/4 and 1K for lambda2/5). In contrast, two primers were designed for the heterologous lambda4 subgroup (1M and 1N), based on sequences published by Vasicek and Leder (1990) and Kabat et al. (1991).
To clone the amplified immunoglobulin gene fragments into expression vectors, a second set of homologous primers (Set 2) containing restriction endonucleases sites was designed. These primers add the recognition sequences of Nco I (5') and Hind III (3') to the amplified heavy chain gene fragments and Mlu I (5') and Not I (3') to the amplified light chain gene fragments. These restriction enzymes have been selected primaryly, (a) because of their low probability to cut within human variable heavy and light chain coding regions. Additionally, (b) they produce overlaps of 4 nucleotides or more leading to optimal cloning efficiency. The enzymes (c) do not depend on methylation and (d) their efficiency in recommendend double digestions is more that 90%. Finally, they were selected for cloning the amplified gene fragments into pSEX or pOPE vector derivatives (Breitling et al., 1991; Duebel et al., 1993).
Material Required
PCR reagents and equipment
Preparation of Reagents
The mRNA and cDNA from lymphocytes should be prepared as described in standard protocols.
PCR-Setup
We propose to use a commercial PCR kit containing a proof-reading polymerase and to set up the reactions as recommended by the manufacturer. Most PCRs are carried out in a 50 microliter reaction. Within this volume, we recommend to use 50-100 ng of cDNA and 25 pmol (2.5 microliter) of each primer (10 pmol/microliter). With the first set of primers 15 separate reactions should be performed. Each variable heavy chain forward primer (1A-C) has to be combined with the constant region IgG or IgM reverse primer (1D, 1E), respectively. Analogous, each kappa- and lambda-light chain forward primer (1F-H, 1I-O) has to be combined with the corresponding constant region reverse primer (1P or 1Q), respectively. The reaction mixtures accomplished with dNTPs, buffer and polymerase should be subjected to 30 PCR cycles using an annealing temperature of 55°C. Analyse the reaction mixtures by electrophoresis using a 1.5% agarose gel. Amplified DNA fragments of approx. 380 to 400 bp should be gel purified and subjected to an analogous second PCR using the second primer set to introduce the restriction endonucleases sites.
References/Publications (2)
Downloads
Q & A's
Customer Reviews
Login